
Ozone Layer Holes: ¿A Threat to Life on Planet Earth? Let's learn a little bit more about the depletion of the Ozone Layer and it's Impact on Planet Earth. Over the past 30 years, we humans have become familiar with terms like the Ozone Layer, and some other concepts such as Ozone Hole aka Ozone Layer Hole or Depletion and Climate Change.
The 80s and 90s were decades in which the consequences of human technological progress and industrialization of, particularly, countries in the Northern hemisphere were brought to the table and discussed.
The negative consequences for life on Planet Earth were crystal clear. Scientists started to study and publish the results of their investigations and the conclusion they reached was inescapable: human activities have a negative impact on the Ozone Layer; they also negatively contribute to the further depletion of the Ozone hole and thus, the rise of Global Warming, which is the main cause of Climate Change all around the globe.
In this article, we’ll explore very briefly the origins of these major problems and we'll dig into some statistics that show us empirically what the current situation is. Besides we’ll explore together what we can do on a daily basis to contribute to the ease of this worrying environmental issue.

Before we start, however, we need to become familiar with some key concepts and words so that we can understand a little bit better how it’s all related. Let’s check them out!
1. Ozone: Ozone (O3) is a highly reactive gas composed of three oxygen atoms. It is both a natural and a human-made product.
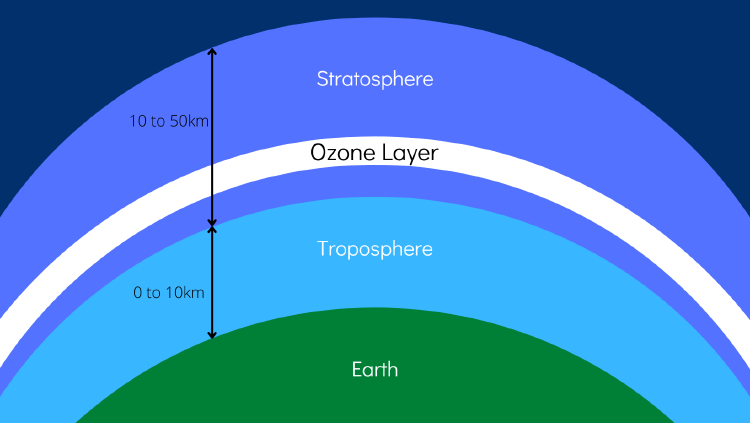
2. The stratosphere: it’s the Earth's upper atmosphere
3. The Troposphere: it’s the Earth’s lower atmosphere.
4. The Ozone Layer: Ozone is found in two different parts of our atmosphere:
5. The Ozone Hole: The term ‘ozone hole’ refers to the depletion of the protective ozone layer in the upper atmosphere (stratosphere) over Earth's polar regions, particularly the Antarctic Pole.
6. Chlorofluorocarbons or CFCs: compounds with chlorine and/or fluorine attached to carbon.
7. Hydrofluorocarbons (HFCs): a group of industrial chemicals primarily used for cooling and refrigeration. HFCs were developed to replace ozone-depleting substances that are currently being phased out under the Montreal Protocol on Substances that Deplete the Ozone Layer.
8. Halons: similar compounds to CFCs with bromine or iodine. These chemicals can remain in the atmosphere for decades to over a century.
9. Climate Change: Climate change includes both global warming caused by human emissions of greenhouse gases and the resulting large-scale shifts in weather patterns.
For further information on this, please check this video from National Geographic on YouTube:
As we previously said, the ozone layer is a natural barrier -composed of “good” ozone- against the UV rays coming directly from the sun. The sun emits electromagnetic radiation at different wavelengths, meaning energy at different intensities and the atmosphere acts like a multi-layer shield that protects Earth from dangerous solar radiation. If it weren’t for the ozone layer, it would be impossible for us human beings to be exposed to the sun at all!. Yap, as crazy as it sounds.
Unfortunately, in the past few decades, scientific studies have proved that human activities and the so-called “progress” in the flourishing of industrialized nations, breakthroughs in technology, military research, among others activities, have a really negative impact on the environment.
How so? It turns out that the extremely high levels of pollution resulting from these human activities alter in the long term the chemical composition of the ozone layer. This produces enormous damage since the chemicals released into the atmosphere provoke a progressive thinning of the ozone layer, which brings about terrible problems for all the living things on our planet.
In the case of human beings, we are more exposed to sun UV and UVB radiation, which most probably translates into skin and eye damage. If you are interested in this topic, please check our article about: Sun and Skin Protection: How To Healthily Enjoy Sun Exposure.
But we are not the only ones who suffer the consequences from the depletion of the ozone layer. UVB radiation affects the physiological and developmental processes of plants. Despite the natural mechanisms developed by plants to cope with this alteration, plant growth may be directly connected to UVB radiation as in the case of changes in plant form, how nutrients are distributed within the plant, etc, etc. This means, in simpler terms, that these changes can have important implications for plants cycles and thus, plants supply.
Animals are no exception to the negative consequences of the thinning of the ozone layer, either and the greatest impact is on sealife. Exposure to solar UVB radiation affects both orientation and motility in phytoplankton (the basis for the existence and survival of marine ecosystems), resulting in reduced survival rates for these organisms. Scientists have demonstrated a direct reduction in phytoplankton production due to ozone depletion-related increases in UVB.

Now, you may be wondering at this point: How did we come so far? If you are willing to continue reading, we´ll briefly explore the answer.
In the 1980s, scientists discovered that the ozone layer was thinning in the lower stratosphere, with particularly dramatic ozone loss in the Antarctic spring (September and October). This great ozone loss in the Poles - both the Arctic and the Antarctic- has become known as “the ozone hole” which is not a hole in literal terms, but it is quite an accurate term since the ozone layer is significantly thinner in these areas.
Scientists also discovered that the depletion of the ozone layer was caused by increasing concentrations of ozone-depleting chemicals: chlorofluorocarbons or CFCs and to a lesser extent halons. These chemicals are not easily expelled from the atmosphere and can stay there for decades to over a century.
Now the thing is that at the poles, CFCs attach to ice particles in clouds. When the sun comes out again in the polar spring, the ice particles melt, releasing the ozone-depleting molecules from the ice particle surfaces. Once released, these ozone-destroying molecules do their job, breaking apart the molecular bonds in UV radiation-absorbing ozone.

Luckily, though, recognition of the harmful effects of CFCs and other ozone-depleting substances led to the Montreal Protocol on Substances That Deplete the Ozone Layer in 1987, a milestone agreement to phase out those substances that has been ratified by all 197 UN United Nations member countries.
If you make it up to this point in the article, you’ll be pleased to find out that numbers and figures in relation to this issue are pretty favorable. Let’s find out!
More than 30 years after the Montreal Protocol, NASA scientists documented the first direct proof that Antarctic ozone is recovering because of the CFC phase-down: Ozone depletion in the region has declined 20 percent since 2005. And at the end of 2018, the United Nations confirmed in a scientific assessment that the ozone layer is recovering, projecting that it would heal completely in the (non-polar) Northern Hemisphere by the 2030s, followed by the Southern Hemisphere in the 2050s and polar regions by 2060.
Monitoring of the ozone layers continues and some results indicate that the recovery may not be as quick and positive as projected. An unexpected and inexplicable drop in the lower ozone layer was registered early in 2018 but the causes are not yet known. Some point out that this may be the result of some or most nations not fully committing to the Montreal Protocol.
It becomes clear that the world is not yet in the clear when it comes to harmful gases from coolants and lubricants, for instance. Some hydrochlorofluorocarbons (HCFCs), transitional substitutes that are less damaging but still harmful to ozone, are still in use. At the same time, developing countries need funding from the Montreal Protocol's Multilateral Fund to eliminate the most widely used of these, the refrigerant R-22. The next generation of coolants, hydrofluorocarbons (HFCs), do not directly contribute to thinning the ozone layer, but they are still fabricated with dangerous greenhouse gases that cause global warming, and therefore contribute negatively to the changing of climate patterns.
This situation with new coolants, refrigerators and lubricants that have a planet-warming effect urged an addition to the Montreal Protocol, the Kigali Amendment, in 2016. The amendment, enforced in January 2019, pretends to cut down the use of HFCs by more than 80 percent over the next three decades. Meanwhile, companies and scientists together are working on climate-friendly alternative products, using less harmful chemicals.
Check this post: Climate Crisis Books: What You Must Read to Know What's happening
Well then, we may ask ourselves a final question: Is there anything we can do on a daily basis to protect the ozone layer?
Although the vast majority of ozone-depleting substances usage is either industrial or commercial, we as individuals can help in some ways:
Learn How to Design a Sustainable Home? 50 Easy Sustainable Home Tips!

According to current legislation (under Title VI of the Clean Air Act) EPA is responsible for developing and implementing programs that protect the ozone layer. You can find programs near your place.
You may be interested at: Sustainable Development Goals (SDG): How to Put Them In Practice?
Let’s continue our efforts to protect the ozone layer from depleting even further!